

Indian Journal of Science and Technology
DOI: 10.17485/IJST/v16i12.2445
Year: 2023, Volume: 16, Issue: 12, Pages: 931-940
Original Article
Nikita Prajapati1, Shalini Chaturvedi2*
1Department of chemistry, Silver Oak Institute of Science, Silver Oak University, Ahmedabad Gujarat, India
2Department of Chemistry, Shri R K Parikh Arts and Science College Petlad, Anand, Gujarat, India
*Corresponding Author
Email: [email protected]
Received Date:21 December 2022, Accepted Date:13 February 2023, Published Date:28 March 2023
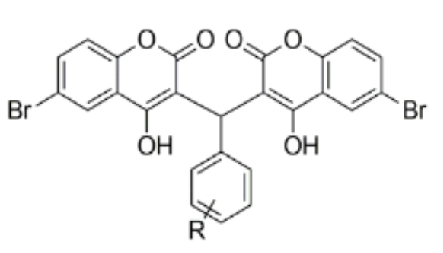
Objective: To synthesize Ni(II) complexes on the base of enrofloxacin with dicumarol derivative and to investigate their antioxidant and antimicrobial potentials. Methods: Ni(II) complexes have been synthesized by classical thermal methods by mixing aqueous metal solutions with ethanol ligands and enrofloxacin in a 1:1 molar ratio. A biologically strong ligand was prepared by refluxing a dicoumarol derivative in aldehyde and ethanol. The structures of the ligands and their nickel complexes were investigated and confirmed by elemental analysis, FT-IR, 1H-NMR, 13C-NMR and mass spectroscopy. The thermal behavior of newly synthesized mixed-ligand Ni(II) complexes was studied by thermogravimetric analysis. Ferric-reducing antioxidant power (FRAP) (antioxidant activity) of all complexes were measured. All the compounds were screened for their antimicrobial activity against E. coli; P. aeruginosa; S. pyogenes; C. albicans and A. niger using Luria Broth dilution method for MIC values. Both the ligand and its complex were screened for antioxidant and antimicrobial activity in vitro. Findings: Antioxidant and antimicrobial activities of complexes were found to be better than the parent ligands used for complex formation. MIC values for complex C5 (-CH3 derivative) were found to be between 40-70 mg/mL and FRAP values was 82.44 mmol/100g, indicative of its ferric reducing antioxidant power. These result show that complex C5 (-CH3 derivative) was found more potential as antimicrobial and antioxidant agent. Novelty : The coordination of metal complex with enrofloxacin (antibiotic) draws a scheme for the synthesis of new drug as well as increasing the activity of enrofloxacin that is currently in use. Current work supports that the activity of antibiotics could be increased by coordination of metal ion complex. There could be a possibility that these complexes allow to reduce the dose of drug introduced into the body.
Keywords: Dicumarol; Antimicrobial; Antioxidant; Thermal Analysis; TGA
© 2023 Prajapati & Chaturvedi. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Published By Indian Society for Education and Environment (iSee)
Subscribe now for latest articles and news.