

Indian Journal of Science and Technology
DOI: 10.17485/IJST/v14i10.1965
Year: 2021, Volume: 14, Issue: 10, Pages: 858-868
Original Article
Anjan Das1*
1Assistant Professor, Department of Physics, Government General Degree College Singur, 712409, West Bengal, India. Tel.: +91 9434527789
*Corresponding Author
Tel: +91 9434527789
Email: [email protected]
Received Date:29 December 2020, Accepted Date:14 March 2021, Published Date:02 April 2021
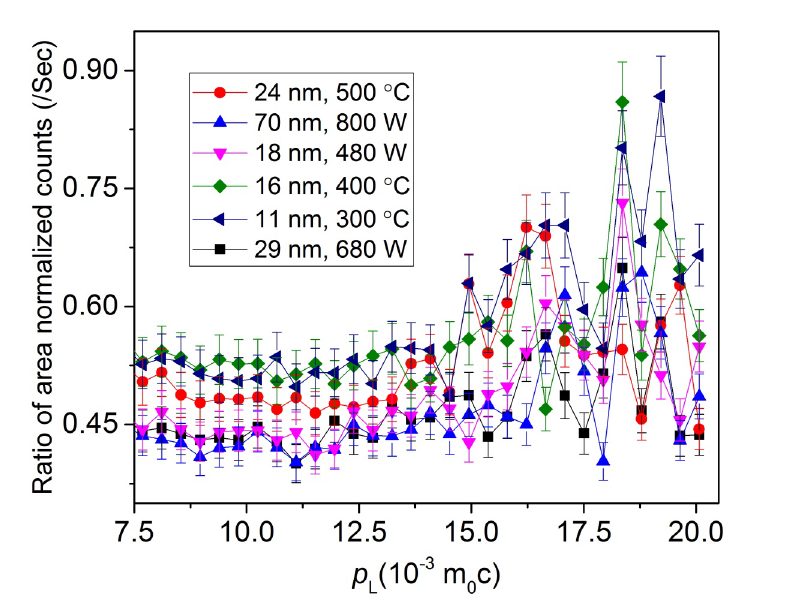
Objective: The objective of this study is to synthesize CdO nanoparticle and nanoflakes both in a single way. Also the purpose of removing defects from semiconductor nanoparticles has been considered. Methods: Very small sized Cadmium oxide nanoparticles (11 – 29 nm) and nanoflakes of average size 70 nm were prepared by sol-gel process. Two solutions of 0.2M CdCl2 and 0.2M NaOH were used with a mixture of 1:2 ethylene glycol and methanol. Two different heating method of dried precipitate was adopted to observe the morphology and defect characteristics. Half of the precipitate was annealed in furnace and rest half in domestic microwave. Findings: The fcc (NaCl-type) structure has been found in the nanostructures with lattice constant varying with annealing temperature. We propose a linear relation for the change in lattice constant. HRTEM pictures show nanoflakes and hexagonal nanoparticles. The ratio curve of coincidence Doppler broadening spectroscopy (CDBS) found defect concentration reduction and agglomeration effect on microwave irradiation in place of furnace annealing. It indicated reduction of defects inside the nanoparticles. The S and W parameter revealed the chemical surroundings and momentum distribution of the vacancy clusters varying with crystallite size. Novelty: Furnace annealing and microwave irradiation showed different effect of surface strain on lattice. Microwave irradiation has been used to reduce defects inside CdO nanocrystals for potential application and coincidence Doppler broadening spectroscopy showed potential to be a very effective tool of defect characterization and electron momentum comparison. Also, this work presents how microwave radiation can be used to prepare nanoflakes and nanoparticles from same reactants without any substrate.
Keywords: Domestic microwave; nanocrystals; positron annihilation; solgel processes
© 2021 Das.This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Published By Indian Society for Education and Environment (iSee)
Subscribe now for latest articles and news.