

Indian Journal of Science and Technology
Year: 2020, Volume: 13, Issue: 19, Pages: 1944-1954
Original Article
Kamal Jaiswal1 , Suman Mishra2 , Anjum Bee3∗
1 Professor, Department of Zoology, Babasaheb Bhimrao Ambedkar University, Lucknow, 226025, India
2 Assistant Professor, Department of Zoology, Babasaheb Bhimrao Ambedkar University, Lucknow, 226025, India
3 Research Scholar, Department of Zoology, Babasaheb Bhimrao Ambedkar University, Lucknow, 226025, India. Tel.: 07905696218
∗Corresponding author:
Anjum Bee Research Scholar,
Department of Zoology, Babasaheb Bhimrao Ambedkar University, Lucknow, 226025, India.
Tel: 07905696218
Email: [email protected]
Received Date:12 April 2020, Accepted Date:23 April 2020, Published Date:18 June 2020
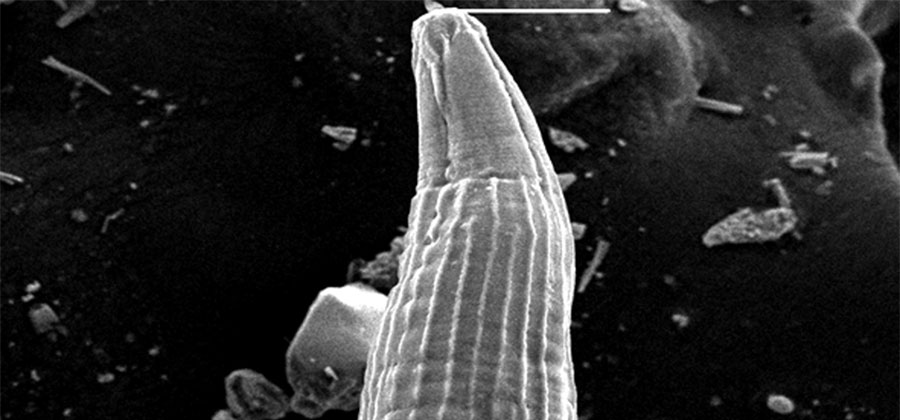
Background/objectives: Ascaridia galli (Schrank) is a well-known nematode parasite of poultry birds. Heavy infection causes an economic loss to the poultry farmers. Information related to its detailed morphological and anatomical structure is inadequate. A study of ultrastructure has revealed the possibility of differentiating the nematodes. Methodology: Ascaridia galli were collected from the domestic fowl (Gallus gallus domesticus) from Lucknow, UP. Scanning electron microscopy was used for the detailed identification of Ascaridia galli. Findings: Scanning electron microscopy exposed that mouth was surrounded with three trilobed lips. Internal rim of each lip was surrounded with fine teeth, and sensory papillae on the outside. Annulations of cuticle occur on the body surface, which is further divided into subannuli. Posterior end of male was pointed and curved having a precloacal sucker and anus. Females had a blunt and straight tail with an anus at ventral side. Caudal papillae and the lateral caudal alae are the tiny outgrowths which surrounds the each side of posterior opening. A sclerotized ring bounded to precloacal sucker was present. These signs of Ascaridia galli attained in the present study could be helpful in the taxonomical studies of nematode worms. Development of control strategies is very essential by understanding the host–parasite relationships. For this purpose, identification of nematode parasite will be very useful. Novelty/ contribution: The present study was designed to understand the morphology of A. galli. Therefore, these markers could be helpful in the taxonomical status of ascaridia nematode worms in domestic fowl.
Keywords: Domestic fowl; A galli; Precloacal sucker; Trilobed lips; Caudal papillae; Scanning electron microscopy
© 2020 Jaiswal, Mishra, Bee. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Published By Indian Society for Education and Environment (iSee)
Subscribe now for latest articles and news.