

Indian Journal of Science and Technology
DOI: 10.17485/IJST/v15i45.1783
Year: 2022, Volume: 15, Issue: 45, Pages: 2492-2499
Original Article
K A Jamal Basha1, Phaneendra Reddy Guddeti2, Tulasi Ramakrishna Reddy Kotte1*
1Solar Energy Laboratory, Department of Physics, Sri Venkateswara University, Tirupati, 517502, Andra Pradesh, India
2Faculty in Physics, Department of Humanities and Sciences, Dr. YSR Architecture and Fine Arts University, Kadapa, 516002, Andra Pradesh, India
*Corresponding Author
Email: [email protected]
Received Date:01 September 2022, Accepted Date:19 October 2022, Published Date:07 December 2022
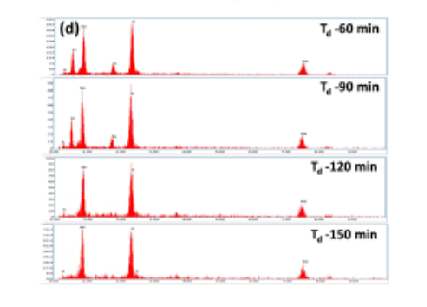
Objectives: To study the influence of deposition time on the physical behavior of NiS films formed by chemical bath deposition (CBD). Methods: Polycrystalline NiS thin films were deposited by using CBD method on glass substrates by varying the deposition time in the range of 60–150 min with the other growth conditions kept constant. The physical properties were measured using an X-ray diffractometer, Scanning Electron Microscopy with Energy Dispersive X-ray analyzer, and Fourier transform infrared spectra. Finally, the optical and electrical properties of the films were analyzed by using UV-Vis spectrophotometer and linear four-probe point method respectively. Findings: The X-ray Diffraction (XRD) studies showed polycrystalline nature of the films with hexagonal structure, confirmed by the Rietveld refinement analysis. The calculated crystallite size varied from 6 nm to 19 nm with the increase in deposition time. The EDS analysis revealed the stoichiometry of Ni and S in the samples. The optical bandgap decreases from 2.06 eV to 1.93 eV with the increase in deposition time. The films synthesized using a deposition time of 120 min at 80 ◦C showed a high electrical conductivity of 48.3 S/cm at room temperature with activation energy of 0.16 eV. Novelty: NiS thin films were deposited by the CBD method using different deposition times varying from 60 min. to 150 min., keeping the bath temperature constant at 80 ◦C for the first time. The Rietveld refinement analysis was the first of its kind, reported on the structural evaluation of NiS layers. These films were formed using eco-friendly materials adding value to the solar cell application.
Keywords: Thin films; Absorber layer; CBD method; XRD; FTIR
© 2022 Basha et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Published By Indian Society for Education and Environment (iSee)
Subscribe now for latest articles and news.