

Indian Journal of Science and Technology
DOI: 10.17485/IJST/v15i48.2092
Year: 2022, Volume: 15, Issue: 48, Pages: 2707-2715
Original Article
P Naresh Kumar Reddy1, D Nagamalleswari2, Dadamiah P M D Shaik3, P Vishnu Prasanth2*
1Department of Physics, Mohan Babu University, A. Rangampet, 517 102, India
2Department of Physics, Sree Vidyanikethan Engineering College, A. Rangampet, 517 102, India
3Department of Physics, Lords Institute of Engineering and Technology, Hyderabad, 500091, India
*Corresponding Author
Email: [email protected]
Received Date:27 October 2022, Accepted Date:28 November 2022, Published Date:24 December 2022
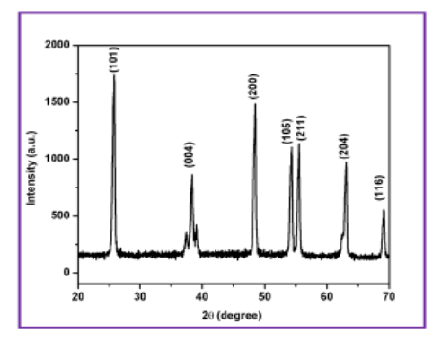
Objectives: To investigate the electrochemical performance of TiO2 (Titanium dioxide) nanoparticles prepared from Ocimum tenuiflorum Plant (OTP) Leaves extract. Methods : cost-effective and eco-friendly green synthesis approach is used to synthesize the TiO2 nanoparticles. XRD (X-ray diffraction) and FESEM (Field Emission Scanning Electron Microscopy) techniques are used to analyse microstructural details. Raman and EDX (Energy Dispersive X-ray spectroscopy) are used to analyse the phase and the chemical composition of the synthesized TiO2 nanoparticles. The nature of chemical bonding as well as the functional groups that the sample contains is identified by using FITR (Fourier Transform Infrared spectroscopy) investigation. The optical band gap of the prepared nanoparticles is estimated by UV-Vis spectroscopic analysis. Finally, Cyclic Voltammetry (CV), chronopotentiometry (CP) and electrochemical impedance spectroscopy (EIS) are used to investigate the electrochemical performance of the produced TiO2 nanoparticles. Findings: The XRD data exhibited that the prepared TiO2 nanoparticles are in a tetragonal structure with an anatase phase and have a crystallite size of 18.6 nm. FESEM images of the TiO2 nanoparticles confirm the smooth surface morphology of spherical grains having an average grain size of 92 nm. The formation of the anatase phase is confirmed by the Raman spectroscopic analysis. The Ti-O-Ti bonds are identified in the sample through the FTIR absorption spectra. The optical band gap of green TiO2 nanoparticles is found to be 3.07 eV and indexed to the anatase phase. Moreover, the better electrochemical performance of the prepared TiO2 nanoparticles is identified from both CV and CP studies in Na2SO4 aqueous electrolyte. At last, the capacitive retention attains up to 65% even after 5000 cycles for the sample prepared from OTP. Novelty: A significant component of this study is the creation of the 3D nanostructured morphology of TiO2 nanoparticles using the green synthesis method which followsthe green chemistry principles. This type of research provides assurance for the protection of rights of the future generations and ecosystems. This type of 3D nanostructure demonstrates superior performance as a supercapacitor electrode, photocatalyst, antibacterial agent, UV-resistant material and solarpowered H2 fuel producer.
Keywords: TiO 2 nanoparticles; Green synthesis; Ocimum tenuiflorum Plant (OTP); Optical band gap; Specific capacitance
© 2022 Reddy et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Published By Indian Society for Education and Environment (iSee)
Subscribe now for latest articles and news.