

Indian Journal of Science and Technology
DOI: 10.17485/IJST/v13i36.1099
Year: 2020, Volume: 13, Issue: 36, Pages: 3762-3771
Original Article
Gayatriben B Patel1*, Kamlesh R Shah1
1Department of Biotechnology, Pramukh swami science, and H.D. Patel Arts College, Kadi,India
*Corresponding Author
Email: [email protected]
Received Date:09 July 2020, Accepted Date:26 September 2020, Published Date:09 October 2020
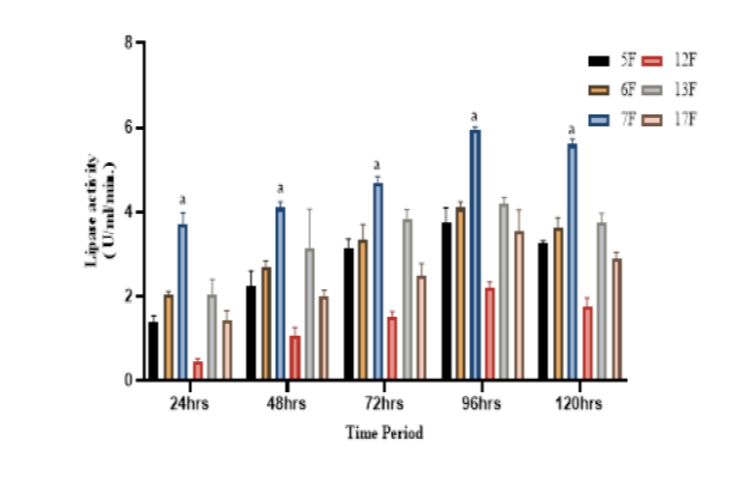
Background/Objectives: The present study was focused to exploit the indigenous strains of fungi isolated from cotton seed soapstock for the production of the extracellular lipase through submerged fermentation technique. Methods/ Statistical analysis: Cotton seed soapstock samples used in the study contains gelatinous oil richer chemical constituents. In addition, their enrichment and diluted materials were used for the isolation of lipase producing microorganisms on tributyrin agar plates. All isolates were lipase positive confirmed by a qualitative plate assay. Quantitative estimation of Lipase production activity was measured spectrophotometrically using pnitrophenyl palmitate (p-NPP) as a substrate. In order to exploit the isolated fungal strain for industrial usage, other cellulase and protease enzymes were tested by plate assay. Morphological and molecular characteristics of selected isolates were studied. Findings: From enriched soapstock samples, a total of 49 cultures were isolated among them 19 fungal isolates were screened for lipase, cellulase, and protease activity qualitatively by plate assay. Out of 19, six fungi were selected based on their lipase activity. Highly potent Fusarium solani 7F had the ability to produce 5.95 U/mL/min. crude lipase whereas Aspergillus niger 13F has 4.2 U/mL/min. after 4days incubation at 30oC. Potent fungi culture was identified by morphological, cultural, and molecular characteristics (18s rRNA gene sequence and phylogenetic analysis) revealed them as Penicillium griseofulvum 5F, Aspergillus flavus 6F, Fusarium solani 7F, Aspergillus niger 12F, Aspergillus niger 13F, and Aspergillus terreus 17F. Novelty: Fungi was the first time reported and isolated from cotton seed soapstock materials. In future studies, this enzyme will be used in the degradation of soapstock and also in the production of biosurfactant from soapstock.
Keywords: Cotton seed soapstock; Fusarium solani 7F; p-nitrophenyl palmitate; Tributyrin agar plates; degradation
© 2020 Patel & Shah.This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Published By Indian Society for Education and Environment (iSee).
Subscribe now for latest articles and news.