

Indian Journal of Science and Technology
Year: 2020, Volume: 13, Issue: 31, Pages: 3150-3159
Original Article
Mac Van Hung1, Vu Anh Tuan1*
1School of Chemical Engineering, Hanoi University of Science and Technology, Vietnam.
Tel.: (+84) 912.911.902
*Corresponding Author
Tel:(+84) 912.911.902
Email: [email protected]
Received Date:10 June 2020, Accepted Date:07 August 2020, Published Date:27 August 2020
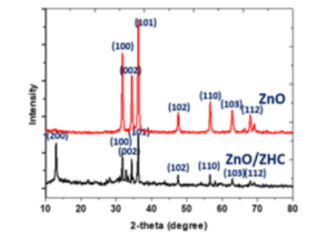
Objective: This study aims to degrade the organic dyes in aqueous medium and low UV intensity by nano-rod ZnO. Methodology: The ZnO was prepared by a facile and fast method, as-synthesized ZnO was characterized by SEM,XRD, N2 adsorption/desorption isotherm, and diffuse reflectance UV-vis. The composition of samples before and after calcination steps was analyzed by XRD. The catalytic performance of ZnO was evaluated by the degradation of dyes in aqueous solution under a low UV light irradiation. Finding: the precursor was completely transformed to ZnO at 400 ◦C. The ZnO was nanorod structure with the crystalline phase of hexagonal wurtzite. The degradation efficiency of ZnO for Janus Green B in 10 min was 96.8%, it was 95.8% in 40 min for Congo Red. The reaction kinetic of photodegradation of dyes was followed by the first-order kinetic model and the photodegradation mechanism of ZnO for dyes was proposed. Application/Improvement: The facile and fast method was developed for the synthesis of nano-rod ZnO, it could be applied for practical application in wastewater treatment.
Keywords: ZnO; dyes; photocatalyst; first-order kinetic; Nano-rod
© 2020 Hung & Tuan.This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Published By Indian Society for Education and Environment (iSee).
Subscribe now for latest articles and news.